SERS substrates
| Code | Description | Substrate | Size, mm2 | Thickness, mm | Active area, mm2 | Price, EUR/pc** |
AUITO-1  (recommended Raman excitation wavelength 785 nm.) (recommended Raman excitation wavelength 785 nm.) | Au nanoparticles coated on ITO glass (all substrate surface is conductive) Conductivity </= 50 Ohms/sq. | ITO glass | 25x25 | 1 | 25x25 | on request |
AUG-2 (recommended Raman excitation wavelength 785 nm.) | Au nanoparticles coated on microscope slide | glass | 25x76 | 1 | 25x20* | on request |
* available custom size.
** 10% discount for each unit when the order quantity exceeds 20 pcs.
- A variety of substrate materials are obtainable.
- Ag nanoparticles coated on a microscope slide are available.
- Different sizes of nanoparticles are possible.
SERS substrates are prepared using laser-based technology. The typical Raman enhancement factor of SERS based on gold nanoparticles is 106-107.
Recommendations for the use of substrates:
- do not exceed 5 mW of laser average power using 50x(NA=0.5) objective during the measurements in Raman microscopy.
- use the substrates within 2 months from the date of the production.
Applications
- Raman spectroscopy
- Electrochemistry
- Chemical identification
- Environment measurements
- Medical diagnostics
SEM MICROGRAPHS OF SERS SUBSTRATES

Fig. 1. a) SEM micrograph of ITO substrate coated with Au nanoparticles; b) and c) SEM images of a glass substrate coated with different size and density of Au nanoparticles.
RAMAN SPECTRA OF SERS SUBSTRATES
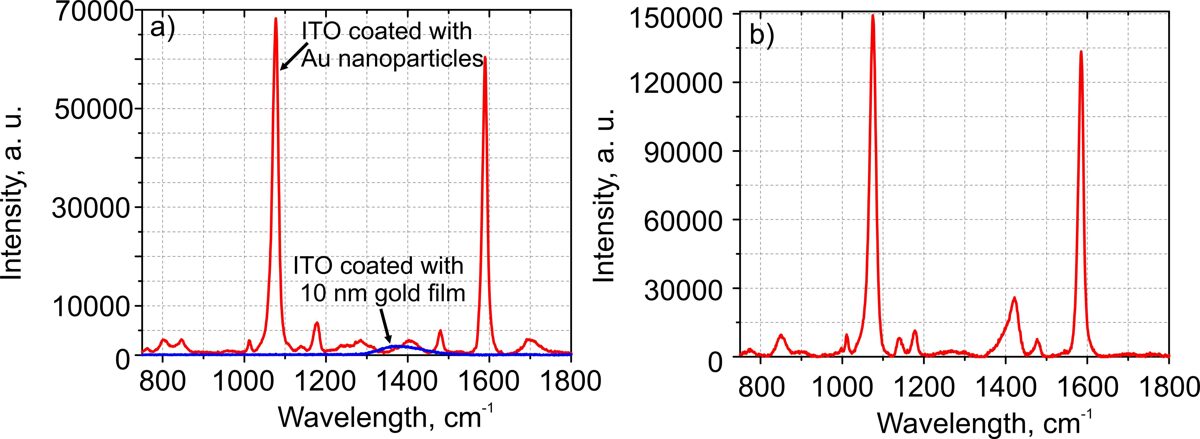
The comparison of Raman spectra of 2-Mercaptobenzoic acid on the ITO glass coated with Au nanoparticles (red curve) and on the ITO glass coated with a 10 nm gold film (blue curve) shown in Fig. 2a.
SERS substrate enhances the Raman scattering light from molecules, capable of detecting trace amounts of chemicals and identifying them based on their unique vibrational characteristics.
Measurement conditions of Raman spectra:
Sample preparation: SERS substrates were immersed in an ethanol solution of 1mM Merkaptobenzoic acid for 16 hours. Afterward, substrates were rinsed in ethanol (96%) and dried at room temperature under slow nitrogen gas flow.
Raman measurement: laser wavelength 785 nm; average power 0.9 mW; 50x objective (NA=0.5); grating 1200 l/mm; integration time 100 s.
Applications for SERS substrates:
- Raman spectroscopy
- Electrochemistry
- Chemical identification
- Environment measurements
- Medical diagnostics
THE PRINCIPLE OF RAMAN SPECTROSCOPY
Raman spectroscopy is a spectroscopic technique where scattered light is used to measure the vibrational energy modes of molecules, although rotational and other low-frequency modes of systems may also be observed.
When monochromatic light is irradiated on molecules, the light is scattered by molecules. Most scattered light has the same frequency with incident light but a small fraction (10-7) of the scattered light has a frequency different from the incident light due to the interaction between oscillation of light and molecular vibration (Fig. 3). Raman shifted frequency (or wavelength) depends on the vibrational state of the molecule. Stokes radiation occurs at a lower frequency (longer wavelength) compared to Rayleigh scattering, whereas Anti-Stokes radiation occurs at a higher frequency (shorter wavelength). The shift in frequency of the inelastically scattered radiation provides the chemical and structural information of the molecule.
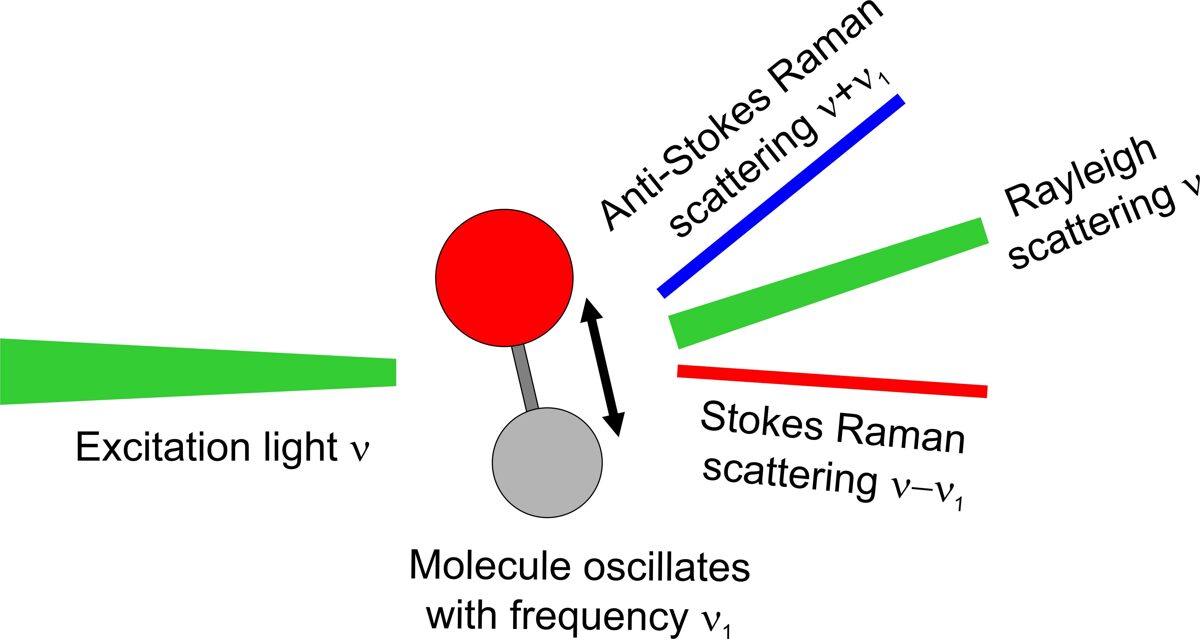
Fig. 3. The principle of Raman scattering.
SURFACE-ENHANCED RAMAN SPECTROSCOPY
Surface-enhanced Raman scattering (SERS) effect deals with the huge amplification of the weak Raman scattering intensity by molecules in the presence of a nanostructured metallic surface (Fig. 4).
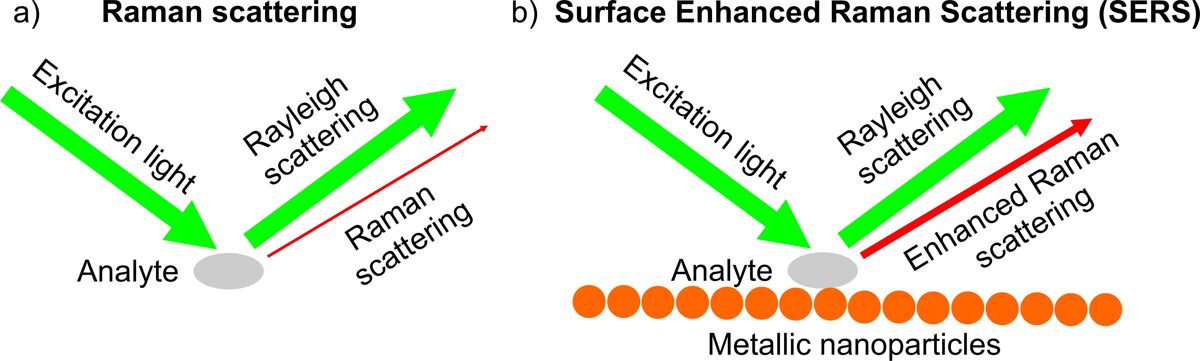
SERS is based on an enhancement of the electric field provided by the coupling of the radiation field with the localized surface plasmons of the metal nanoparticles. The electromagnetic enhancement for SERS depends on the structure of the supporting plasmonic material and theoretically can reach factors of 1010 - 1011.
